Your quote list is empty
Add products from our catalog to request a quote.
Request Submitted!
Thank you for your interest. Our team will review your request and get back to you with a detailed quote shortly.

We are proud to announce that SureDuty has successfully passed its ISO 13485:2016 recertification audit with zero non-conformities. This achievement underscores our unwavering commitment to maintaining the most rigorous quality management systems in the medical device industry. The audit, conducted by a leading international body, covered all aspects of our design, development, production, and distribution processes. This certification provides our partners with the confidence that SureDuty products consistently meet the highest safety and quality standards required in global markets.

Packaging Volume: :1gallon,5Lt,10Lt Dilution Ratio: :35x, 36.83x, 45x Glucose Options: Available with or without glucose; specific concentrations detailed in each formula.
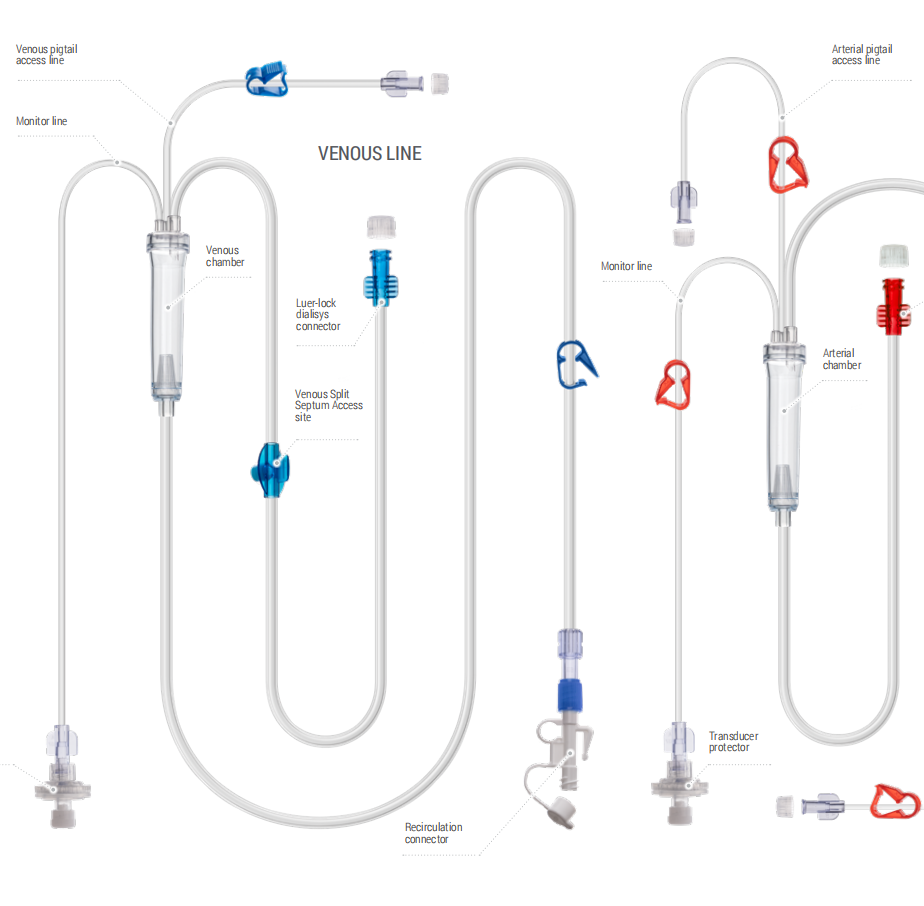
Pump Tubing Sizes: 6.4 × 9.8mm, 8 × 12mm (OD) Priming Options: IV set, Spike, or without priming set Sterilization: EO/GAMMA Sterilized

Acid Concentrate: Contains sodium chloride, potassium chloride, calcium chloride, and acetic acid. Bicarbonate Concentrate: Sodium bicarbonate powder Mixing Ratio: As per dialysis machine or clinical protocol.: Packaging Options: 5 kg or 25 kg packs or customized.:
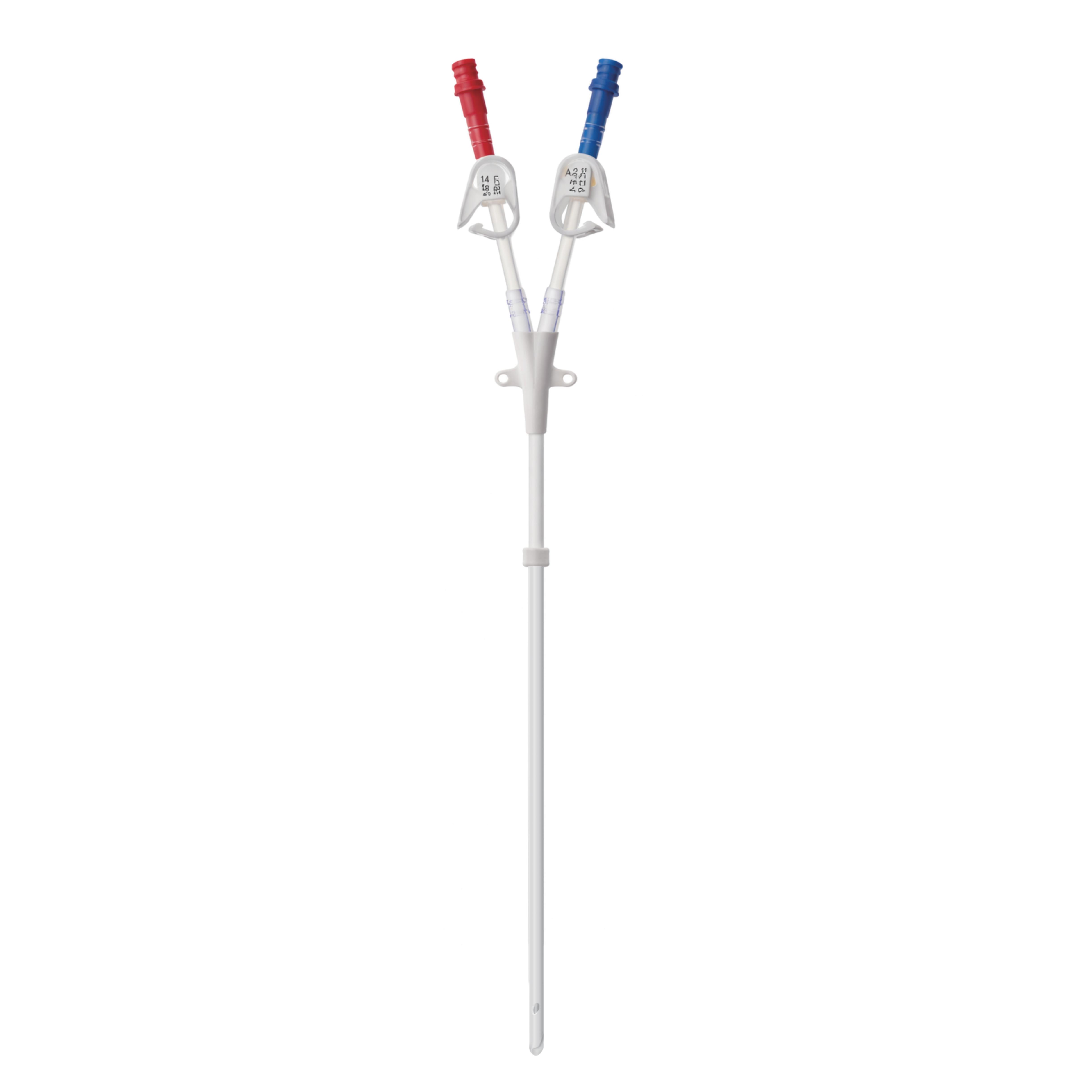
Catheter Sizes: 14.5Fr, 15Fr Lumen Configuration: Dual Lumen Catheter Length: 15 cm -50cm Shape of extension tubes: Straight, Pre-Curved Shape of distal tips: Step type, Symmetric type
Jane Doe is a valued contributor at SureDuty Medical, specializing in global brand strategy and market expansion for renal care solutions.
Add products from our catalog to request a quote.
Thank you for your interest. Our team will review your request and get back to you with a detailed quote shortly.